Primary Biliary Cholangitis Treatment Market to Reach US$ 2.72B by 2033
Rising prevalence of liver disorders, advanced therapies, and increasing awareness drive demand for effective primary biliary cholangitis treatments globally.
The market shows robust growth potential due to innovation in therapeutic options, increased diagnosis rates, and patient awareness, making it attractive for pharma investment and strategic expansion.”
AUSTIN, TX, UNITED STATES, February 26, 2026 /EINPresswire.com/ -- — DataM Intelligence
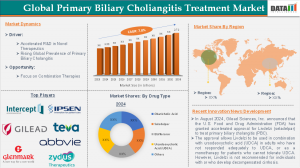
The Global Primary Biliary Cholangitis Treatment Market size reached US$ 1.33 billion in 2024 and is expected to reach US$ 2.72 billion by 2033, growing at a CAGR of 7.8 % during the forecast period of 2025-2033.
The strong 7.8% CAGR is driven by rising PBC prevalence and innovative therapies, such as the U.S. FDA’s accelerated approval of Ipsen’s Iqirvo® and Gilead’s Livdelzi, plus increased clinical trials and collaborations (e.g., CymaBay–Kaken in Japan) expanding treatment access in the US and Japanese markets.
Download your exclusive sample report today: (corporate email gets priority access):https://www.datamintelligence.com/download-sample/primary-biliary-cholangitis-treatment-market
Key Industry Development-
United States: Key Industry Developments
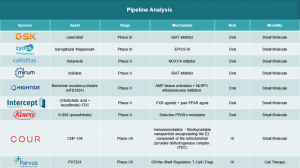
▶ February 2025: Gilead Sciences received FDA clearance for expanded use of seladelpar (Livdelzi) as a second-line therapy for Primary Biliary Cholangitis (PBC) patients with inadequate response to UDCA, demonstrating significant ALP reduction and pruritus relief in clinical trials.
▶ November 2025: Gilead announced positive Phase 3 data for Livdelzi, confirming sustained biochemical response and itch reduction over 12 months, positioning it as a key option to slow PBC progression.
▶ July 2024 (noted for context): Parvus Therapeutics gained FDA Orphan Drug Designation for PVT201, an investigational therapy targeting PBC fibrosis, advancing Phase 1 trials.
Japan/Asia Pacific: Key Industry Developments
▶ February 2025: European seladelpar approval (Gilead) influences Asia-Pacific pipelines, but no Japan-specific launch; regional focus remains on UDCA and emerging fibrates like bezafibrate.
▶ Ongoing 2025: Japanese firms monitor global PBC innovations like saroglitazar in trials, aligning with PPAR agonist trends for second-line PBC management.
Strategic Mergers and Acquisitions:
In April 2025, Johnson & Johnson completed its acquisition of Intra‑Cellular Therapies for approximately USD 14.6 billion, acquiring control of the FDA‑approved drug Caplyta and additional clinical programs for psychiatric and neurologic conditions, strengthening its CNS portfolio and expanding its therapeutic reach in mental health.
In late October 2025, Novartis AG entered into a definitive agreement to acquire Avidity Biosciences for around USD 12 billion, broadening its rare disease pipeline with proprietary RNA‑based technology focused on neuromuscular and other high‑unmet‑need disorders, in a strategic move to diversify beyond existing blockbusters.
Announced in October 2025, Novo Nordisk A/S agreed to purchase Akero Therapeutics for up to USD 5.2 billion, gaining access to efruxifermin (EFX) an FGF21 analogue addressing metabolic and liver disease as part of its long‑term expansion in metabolic‑disease therapeutics.
In September 2025, Roche disclosed plans to acquire 89bio in a transaction valued at up to USD 3.5 billion, securing the rights to pegozafermin, a Phase III‑ready therapy for metabolic dysfunction‑associated steatohepatitis (MASH), reinforcing its cardiovascular‑renal‑metabolic portfolio.
July 2025 saw Sanofi complete its acquisition of Blueprint Medicines for approximately USD 9.5 billion, bringing Ayvakit and a pipeline of KIT‑targeted rare disease therapies under its umbrella to enhance its immunology and rare disease offerings.
Major Players:-
Intercept Pharmaceuticals | Gilead Sciences | Ipsen Biopharmaceuticals | Teva Pharmaceuticals USA | Glenmark Pharmaceuticals | AbbVie | Aden Healthcare | Zydus Therapeutics | COUR Pharmaceuticals | Kowa Company | Mirum Pharma | Parvus Therapeutics | GSK | Strides Pharma Science | Calliditas Therapeutics
Intercept Pharmaceuticals, Inc. – Intercept holds the largest share (around 36% of the PBC market) due to its flagship FXR agonist Ocaliva (obeticholic acid), the first approved second‑line therapy for PBC. Its focused liver disease portfolio and early regulatory leadership have solidified strong global adoption.
Gilead Sciences, Inc. – Gilead leverages seladelpar (Livdelzi/Lyvdelzi) to capture a growing segment of the PBC market, particularly after accelerated approvals in the US and EU. Its broad biopharma infrastructure and investment in liver disease R&D contribute to increasing market penetration.
Ipsen Biopharmaceuticals, Inc. – Ipsen has secured a key share through Iqirvo (elafibranor), the first new PPAR therapy approved for PBC in years. This approval, along with expansion in rare liver disease research, enhances its competitive position in both US and EU markets.
Teva Pharmaceuticals USA, Inc. – Teva strengthens the market through consistent supply of ursodeoxycholic acid (UDCA) generics, the longstanding first‑line PBC treatment. While its share is smaller than novel biologics, its global scale and generics portfolio help sustain broad baseline therapy usage.
GSK plc. – GSK participates with emerging pipeline assets and combination PBC regimens, bolstered by immunomodulatory and metabolic targets. While its current share is moderate, its diversified global presence and clinical investments position it to grow as novel therapies gain traction.
"Secure your 30% discount - get this report before the offer expires."
:https://www.datamintelligence.com/buy-now-page?report=primary-biliary-cholangitis-treatment-market (Purchase 2 or more Repots and get 50% Discount)
Market Segmenatation-
1. Drug Type Segmentation:
In both the U.S. and Japan, the PBC treatment market is segmented primarily by drug type, with ursodeoxycholic acid (UDCA) remaining the dominant first‑line therapy due to its wide use and established clinical profile. Secondary drug segments include obeticholic acid for UDCA non‑responders and emerging treatments such as seladelpar and elafibranor that target novel pathways like FXR and PPAR to address unmet needs from standard therapy. This segmentation reflects how different classes of medications contribute to treatment uptake and revenue generation within each country’s therapeutic landscape.
2. Age‑Based Segmentation:
The PBC treatment market is divided into age groups to capture differences in treatment demand across population cohorts. In both the U.S. and Japan, the adults (18–64 years) segment represents the majority of treated patients thanks to earlier diagnosis and active management, while the geriatric population (65+ years) is growing rapidly due to demographic ageing and increased disease incidence among older individuals. This segmentation helps forecast how changing age demographics will influence market size and drug utilization patterns.
3. Distribution Channel Segmentation:
Segmentation by distribution channel (e.g., hospital pharmacies, retail pharmacies, and online pharmacies) reveals how patients obtain PBC therapies in each market. In the U.S., hospital and specialty pharmacies play key roles due to treatment complexity and specialist prescription practices, whereas retail and online pharmacy channels are increasingly relevant for long‑term medication adherence and patient convenience. Japan also sees strong hospital pharmacy involvement due to structured healthcare and screening programs that maintain high adherence to prescribed therapies.
Market Drivers:-
• Increasing Prevalence of PBC and Early Diagnosis Rates: Rising incidence of PBC particularly among women aged 40+ is boosting demand for both first‑ and second‑line therapies in the U.S. and globally. Expanded screening protocols and broader clinical awareness are leading to earlier treatment initiation and larger diagnosed patient pools.
• Advancements in Novel Therapeutic Options and R&D: Significant R&D progress in FXR agonists (e.g., obeticholic acid), PPAR modulators (like elafibranor), and immune‑modulating agents is expanding clinical choices. This innovation fuels adoption of more effective, targeted therapies that improve biochemical responses over traditional UDCA alone.
• Strong U.S. Clinical Trial Activity and Treatment Adoption: The United States has seen a surge in PBC clinical trial density and an increased uptake of next‑generation therapies; FXR agonists alone have grown 22 % year‑over‑year usage within major hepatology centers, strengthening the U.S. market pipeline and therapy access.
• Supportive Regulatory and Orphan Drug Frameworks: Regulatory incentives, such as orphan drug designation and priority review programs by agencies like the U.S. FDA and Japan’s PMDA, are accelerating approval timelines and improving patient access to new PBC treatments. These frameworks lower developer risk and encourage biopharma investments in rare liver diseases.
• Healthcare Infrastructure and Awareness Efforts in Japan: Japan’s robust healthcare system and heightened disease awareness initiatives have led to early detection and high treatment adherence (> 80 %) among diagnosed patients. Government support for autoimmune and cholestatic liver conditions continues to grow, reinforcing therapy uptake.
Regional insights:-
U.S. (United States): 46% (Largest regional share, driven by high prevalence, advanced healthcare infrastructure, strong clinical trial activity, and early adoption of novel therapies such as FXR agonists and second‑line treatments).
North America (excluding U.S., inclusive of U.S. & Canada overall): 44% (Dominates the global market; robust reimbursement frameworks and specialized hepatology networks support growth).
Note: U.S. comprises the majority of this share; for strict U.S. focus see above.
Japan: 3.5% (Smaller but steadily growing share of the global PBC market reaching about USD 98 M in 2024 supported by high disease awareness, established screening, and increasing treatment uptake).
Asia Pacific (including Japan): 16% (Includes Japan, China, India and other emerging markets; fastest growth trajectory due to rising diagnosis rates and expanding healthcare access).
Get Customization in the report as per your requirements:https://www.datamintelligence.com/customize/primary-biliary-cholangitis-treatment-market
Conclusion:
The primary biliary cholangitis treatment market is poised for steady expansion, fueled by rising disease prevalence, improved diagnostic capabilities, and continuous innovation in therapies. Healthcare providers and pharmaceutical firms can leverage this growth by focusing on patient-centric solutions, novel drugs, and strategic partnerships to address unmet medical needs while enhancing global treatment access.
Related Reports:-
1. Liver Disease Treatment Market
2. Ursodeoxycholic Acid Market
Sai Kumar
DataM Intelligence 4market Research LLP
+1 877-441-4866
email us here
Visit us on social media:
LinkedIn
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.